Our 95% Forensic Mitochondrial DNA Analysis Success Rate makes us the Gold Standard
Your FIRST STOP for challenging forensic DNA samples.

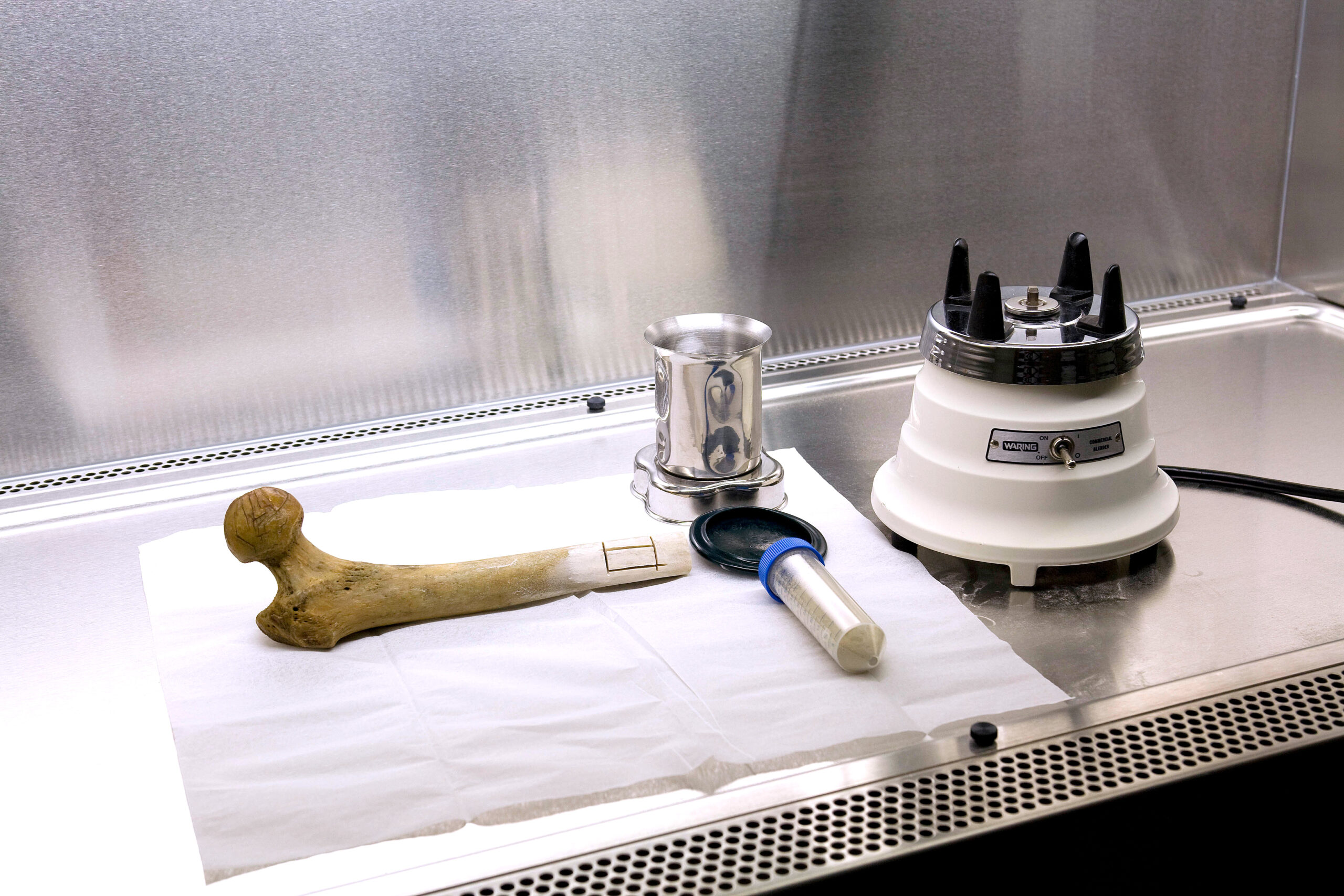
Mitochondrial DNA Analysis is Our Only Focus
The new Mitotyping Technologies is solely devoted to the forensic applications of mitochondrial DNA (mtDNA) analysis. We are one of the oldest and most experienced group of scientists devoted to this forensic and anthropological DNA specialty, having worked on criminal, civil, exoneration and federal cases in 50 states and 13 foreign countries and have testified over 200 times in mtDNA cases for both the prosecution and defense teams.
Global Scope
Mitotyping Technologies provides its mtDNA analysis services throughout the world. After
25+ years
we have serviced
13 countries
working on
1000+ cases
and we are quite excited to offer our superior techniques to your repertoire.
Challenge Us With Your Most Difficult Evidence!
Ancient DNA Methodology
Mitochondrial DNA Analysis of shed hairs and hair fragments is our specialty. We have a 95.5% success rate in developing profiles from hairs of all ages and sizes. For highly degraded samples, we offer an “ancient DNA” approach. Each sample is analyzed individually to account for its specific analytical needs.
Learn our peer-reviewed article in Investigative Genetics.


Proprietary Extraction
Our proprietary extraction techniques have a proven success rate of 95.5% with hair samples as small as 2mm, degraded bone and teeth evidence…so the next time you are faced with challenging evidence, challenge us! We will handle your case in the most expeditious manner with the leading scientific know-how!
Can You Use Species Testing to Distinguish Between Human and Animal DNA on Medical Equipment?
That’s exactly the kind of challenge brought to Mitotyping during a study on whether single-use medical tools can be safely cleaned and reused.
As part of the quality control testing, non-human blood was used to inoculate equipment, followed by an assessment of cleaning effectiveness, which is where Mitotyping can step in…
With more than two decades of experience, we’ve partnered with the medical field to evaluate cleaning methods using species testing and resolve complex contamination cases.
Reach out to learn how we can support your species testing needs!
Did You Know?

Degraded Skeletal Remains
Mitochondrial DNA analysis is typically applied to degraded skeletal remains and telogen or rootless hairs. Data on the application of the method to very small hairs less than 0.5 cm from an age-matched and challenged sample set are lacking.

Mystery of Texas Outlaw Unraveled
Outlaw Wild Bill Longley twice cheated the noose, but popular Texas folklore that he got away a third time is proved wrong with help from Mitotyping Technologies.

We Recover mtDNA from 95.5% of Hairs
Please see our January 2005 publication in Journal of Forensic Sciences about our five years of casework experience with hair analysis. Our protocols for working with degraded DNA give us a superior recovery rate for mtDNA in challenged samples.
